The Caspian Sea is the largest enclosed inland waterbody on our planet, variously classed as the world’s largest lake or a full-fledged sea. Being the residue of ancient seas, the Caspian Sea is completely isolated from oceans now. It is a nondrainage brackish waterbody with profound seasonal and multiannual level oscillations. Because of several marine transgressions and regressions, which have connected the Caspian to other bodies of water, the history of theCaspian biota is dynamic: periods of faunal isolations and species extinctions alternated with species invasions. The present fauna comprises nearly 46% of Caspian endemics (species found only in the Caspian) and 20% endemics of the Ponto–Caspian region (species found only in the Caspian and the Dniepr and Ural watersheds). In recent years, the invasion of a few species decreased the endemism percentage, but it is obviously still significant. The Caspian endemics fall into four groups: (1) species of Tethyan origin (e.g. Sturgeon, Gobiid and Clupeid Fish); (2) species originating from brackish Sarmatian or Pontic Lakes, (e.g. Onychopod Cladoceran Crustaceans); (3) opportunistic freshwater species (most Rotifers, the non-Onychopod Cladoceran Crustaceans, most Cyprinid Fish); (4) a few invaders of northern (Baltic and White Seas) origin, that show few signs of speciation (the Copepod Crustacean Limnocalanus grimaldii, several Mysid Shrimpss, the Caspian Salmon, and the Caspian Seal). These endemic Animals together with freshwater species and species of Atlantic–Mediterranean and Arctic origin represent the current biodiversity of the Caspian Sea.
In the last century, the Caspian Sea was actively studied, but after the disintegration of the USSR, most of the research ceased. However, some of the Caspian species did not get due consideration even in favorable times. This particularly applies to Sponges (phylum Porifera). The endemic Caspian Sponges were first described by Oscar Andreevich Grimm in the 1870s. He reported four species: Reniera flava, Amorphina caspia, Metschnikowia tuberculata, and Metschnikowia intermedia. Later, Voldemar Czerniavsky described a fifth species, Amorphina protochalina, based upon material collected during the Ivashincev expeditions in the 1850s and 1860s. In 1880 Władysław Dybowski proposed to synonymize Reniera flava and Metschnikowia tuberculata, and detailed descriptions of the three further Metschnikowia species based upon, apparently, Grimm’s material. In 1962 Vladimir Koltun reviewed Grimm’s collection and joined Reniera flava, Metschnikowia tuberculata, and Metschnikowia intermedia into the a single species, Metschnikowia tuberculata, although he did not find the original holotypes (specimens from which the species were described). Koltun did succeed in finding the holotype of Amorphina protochalina and suggested it to be an aberrant form of Metschnikowia tuberculata. The only found fragment of Amorphina caspia was regarded by him as not fitting the original description. Thus, the two Amorphina species were considered to be doubtful and requiring confirmation. In a later comprehensive taxonomic revision of Sponges, carried out by Renata Manconi and Roberto Pronzato in 2002, the Caspian sponges became considered as an endemic family with only one species Metschnikowia tuberculata.
Most of the early studies on Caspian Sponges regarded them of being of marine origin. Manconi and Pronzato, however, placed the family Metschnikowiidae within the Freshwater Sponges, suborder Spongillina (now order Spongillida). However, Christine Morrow and Paco Cárdenas subsequently proposed the Metschnikowiids to be reallocated to marine order Haplosclerida, based on their brackish habitat and morphological affinities with Janulum, a marine Haplosclerid.
Thus, given the fact that the Caspian fauna includes species of both freshwater and marine origin, the evolutionary history of Caspian Sponges remained unknown, as did their actual diversity and ecology because it has been a long time since they have been seen alive by biologists.
In a paper published in the journal ZooKeys on 24 February 2020, Agniya Sokolova of the NK Koltzov Institute of Developmental Biology and the AN Severtsov Institute of Ecology and Evolution of the Russian Academy of Science, Dmitry Palatov, also of the Severtsov Institute of Ecology and Evolution of the Russian Academy of Science, and of the Faculty of Biology at Moscow State University, and Valeria Itskovich of the Limnological Institute of the Siberian Branch of the Russian Academy of Science, present the first photos of live Caspian Sponges and their spicules, and as well as a genetic analysis revealing their phylogenetic position.
The material upon which Sokolova et al.'s study is based was collected in the vicinity of Aktau town, Kazakhstan, in September 2018. Specimens were gathered by SCUBA diving, snorkeling or by turning over littoral stones. Sponges were carefully detached from rocks and lower parts of large stones by forceps or were collected with the substratum (Mytilus aggregations). Specimens were fixed in 96% ethanol and RNA-later. When possible, Sponges were photographed in situ before collection. For comparative purposes, specimens of Metschnikowia tuberculata from museum collections (the Zoological Institute of the Russian Academy of Science and the Zoological Museum of Moscow University were also investigated.
For scanning electron microscopy, spicules were purified with potassium dichromate solution and mounted on a stub according to the classical method. Measurements of length and width of spicules were taken with light microscopy.
Total genomic DNA extraction was performed using the InterLabService RIBO-sorb RNA/DNA extraction kit. The 676 base-pair fragment at the 5’ end of the CO1 gene was amplified and sequenced using universal barcoding primers. ITSs were used as a main marker with the best resolution since many species of Freshwater Sponges have the identical COI sequences. Previously described primers were used for amplification of ITS1, 5.8S rDNA and ITS2. Polymerase chain reaction amplifications of ITS1 and ITS2 were performed on a Bio-Rad DNA Engine Dyad thermal cycler using Evrogen 5*ScreenMix. The cycle parameters were initial denaturation at 94°C for 120 seconds, followed by 35 cycles of denaturation at 94 °C for 30 seconds, annealing at 55°C for 30 seconds and extension at 72°C for 120 seconds, followed by a final extension of 8 minutes at 72°C. Each polymerase chain reaction product was purified by electrophoresis in 0.8% agarose gels and eluted by freezing and thawing. Sequencing of both strands of each polymerase chain reaction product was carried out by Syntol using a BIG DYE 3.1 terminator mix on an ABI 377 Sequencer. Chromatograms were analyzed using BioEdit 5.09. All sequences have been deposited with GenBank with the accession numbers MK659927–MK659935 (ITS1 and ITS2) and MN431221–MN431229 (CO1). The assignment of the sequences obtained from Porifera was performed using the BLAST software program. Sequences were initially aligned using ClustalW 1.7 under default parameters including all available sequences of ITS1 and ITS2 of Freshwater Sponges available from GenBank, with mandatory manual correction. Phylogenetic trees were constructed using the maximum likelihood method and Bayesian inference, as implemented in MEGA 5 and MrBayes 3.1.2.
Trochospongilla latouchiana was used as the outgroup for ITS sequences because early branching of this genus among Spongillida has been shown in previous phylogenetic reconstructions. This is the most distant taxon of sponges whose ITS sequences are able to be aligned with the sequences of metschnikowiids. Vetulina stalactites was used as the outgroup for the CO1 sequences as the closest marine group.
The World Porifera Database was used for checking statuses of the taxa under discussion.
Sponges were abundant in the studied depths (0.5–5 m) but preferred hidden places. A total of 41 sponges were collected; nine of them were sequenced.
Sokolova et al. observed sponges of highly variable appearance: crusts of yellow, blue, green or several/transitional colours and bright-yellow spheres, 2-7 cm in diameter. The Sponges of spherical shape were always of yellow colour, while the encrusting Sponges were highly variable in colour. Oscula (large openings to the outside through which the current of water
exits after passing through the spongocoel, having been absorbed through
much smaller dermal pores) were clearly noticeable, quite regularly arranged, sometimes slightly raised above the surface. In some cases, exhalant canals of star-like structure were seen. One of the encrusting sponge morphs differed from others by having smaller oscula, large body area (up to 70 cm²), reduced thickness (3–4 mm) and distinctive faded-green colour.
Thus, two stable morphs could be distinguished among others: thin faded-green encrusting Sponges, and spherical bright-yellow Sponges.
In situ photos of Metschnikowia tuberculata. (A) Globular morph, (B)–(E) encrusting morphs. Scale bars:2 cm (A), (E); 1 cm (B), (C), (D). Sokolova et al. (2020).
The yellow globular morph and faded-green encrusting morph occupied exclusively shaded areas of rocks and lower surfaces of large stones at a depth of 2 m and more.Encrusting sponges of other colors were observed on less-shaded substrata as well as on shaded surfaces at a depth of 50 cm and more.
Spicules were 126–175 μm × 3–7.5 μm, slightly curved or almost straight oxeas (spicules pointed at both ends), from finely spiny to greatly spiny. Sponges of different shapes and colors have no significant distinctions in spicule characters, although oxeas of the yellow globular morphs are generally spinier than in encrusting Sponges. Size and density of spicule spines vary within a specimen.
Skeleton complement of Metschnikowia tuberculata. (A)–(C) Spicules of encrusting Sponges, (D) spicules of a globular Sponge, (E) skeleton arrangement of a globular Sponge, (F) skeleton arrangement of an encrusting Sponge. Scale bars: 30 μm (A)–(D) whole spicules; 2 μm (A)–(D) magnified parts; 250 μm (E), (F). Sokolova et al. (2020).
Oxeas constitute paucispicular ascending tracts, sometimes organized in quite regular anisotropic reticulation. The degree of regularity highly varies, and it is more prominent in peripheral parts of sponges. In encrusting forms, tracts protrude outward from the Sponge surface. In globular Sponges Sokolova et al. observed a smoother surface with only single spicules piercing the surface layer.Spongin is sparse; the basal spongin plate was not found in the collected Sponges.
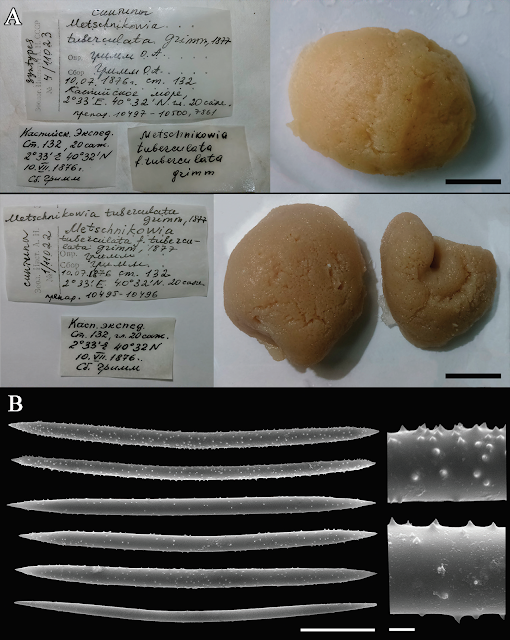
The only existing museum collection of the Caspian sponges includes spirit specimens and slide preparations. This collection was mainly composed of sponges gathered by Grimm in the 19th century, but now the majority of his material is apparently, lost. There are also some sporadic specimens from later expeditions. The wholly preserved Sponges are often of spherical shape and look identical to the globular yellow sponges collected by Sokolova et al. Their spicule composition has no general differences with spicules of sponges from the Aktau vicinity, but their oxeas have larger spines distributed more densely. Spicule size of the museum Sponges varies between specimens, but some of them match with Sokolova et al.'s samples. Numerous slide preparations (made by Vladimir Koltun in the mid-20th century) allow for the estimation of the diversity of spicule characters. The shape of spicules varied from stout, greatly spiny oxeas to thin, fusiform oxeas that bear minute spines.
Museum specimens of Metschnikowia tuberculata. (A) General view of the three syntypes and their labels. Scale bar 1 cm. Zoological Institute of the Russian Academy of Science, specimen №11023. Collector: Oscar Andreevich Grimm. 10.07.1876, station 132; coordinates 2°33'E, 40°32'N, depth 20 fathoms (42.6 m). Associated slides: 10497–10500 (upper specimen) and 10495–10496 (lower specimens). (B) Spicular complement of Metschnikowia tuberculata var. intermedia, Zoological Museum of Moscow University, specimen №251. Scale bars 30 μm (whole spicules) and 2 μm (magnified part). *The longitude is counted from Baku, Azerbaijan, i.e. 2°33'E is actually 52°23'E. Sokolova et al. (2020).
COI sequences of nine sponge samples of different colors and shapes were obtained. All sequences (MN431221–MN431229) were identical and have a length of 676 base pairs. A BLAST analysis revealed that the obtained sequences are most similar to the Freshwater Sponge Ephydatia fluviatilis and differ from its sequence by one nucleotide substitution. The obtained sequences were aligned with available GenBank sequences of Spongillida and Vetulina stalactites (the closest marine relative of Spongillida) and resulted in a 487 base pair alignment, in which 28 characters were available for phylogenetic analyses. Phylogenetic reconstructions based on CO1 data obtained with Bayesian inference and maximum likelihood had similar topologies with poorly resolved phylogenetic relationships. Mean genetic distance between Metschnikowia tuberculata and other Freshwater Sponges was 1% and between Metschnikowia tuberculata and Vetulina stalactites was 10%.
ITS1 and ITS2 sequences were obtained from the same specimens. All sequences (MK659927–MK659935) were identical and have length 751 bp. A BLAST analysis revealed that the obtained sequences are most similar to the freshwater sponge Ephydatia fluviatilis and other Spongillida. The obtained sequences were aligned with available GenBank sequences of Spongillida and resulted in an 873 base pair alignment, in which 437 characters were available for phylogenetic analyses. Marine Sponges were not included in the analysis due to the high variability of ITS spacers making the alignment impossible. Phylogenetic reconstructions obtained with Bayesian inference and maximum likelihood had generally similar topologies, but the Bayesian inference-tree shows higher support.
Bayesian phylogenetic tree based on comparisons of 873 bp of ITS 1 and ITS 2 sequences of Spongillida. Nodes are characterized by Bayesian posterior probabilities (%) followed by bootstrap percentages; a (–) indicates that a particular analysis supported the node at less than 50%, or supported an alternative phylogenetic arrangement in maximum likelihood tree. Trochospongilla latouchiana (Spongillidae; GenBank EF151955) was used as the outgroup. Scale bar denotes substitutions per site. Sokolova et al. (2020).
The analyzed Caspian sponges form a clade with Ephydatia syriaca, Ephydatia fluviatilis, and Cortispongilla barroisi. This clade is included in the common clade with Racekiela sp. and Heterorotula multidentata. All Lubomirskiidae form a strongly supported monophyletic clade with Ephydatia muelleri as a sister species. Sokolova et al.'s results also support monophyly of the genus Eunapius. Radiospongilla is paraphyletic to all other species of Spongillidae.
During its existence, the Caspian Sea has repeatedly reconnected with the ocean. Now isolated, this waterbody retains typical marine features, such as characteristic water circulation, the structure of the water mass, hydrochemical properties, production of organic matter in the pelagic zone, geomorphological structure and distribution of organisms. On the other hand, the Caspian water is characterized by lower salt concentration (12–13‰ in the middle and southern parts) and modified salt composition. Having such a set of features, the Caspian became a cradle of specific fauna, partly of freshwater and partly of marine origin. Some Caspian invertebrates cannot be confidently referred to as animals of either origin, and Sponges were among them. The Caspian Sponges were known to have typical marine habitus, but possess spicules similar to those of Freshwater Sponges. In addition, their distribution within the sea corresponded rather with the hypothesis of marine origin because Sponges were found mainly in the Middle Caspian; these waters are much saltier than fresher North Caspian, where Sponges were almost absent. Based on skeleton features, the Caspian Sponges were considered as a freshwater monotypic family Metschnikowiidae. Based on genetic data, Sokolova et al. have shown that the Caspian Sponge Metschnikowia tuberculata indeed has a freshwater origin.
Sokolova et al. observed two stable morphs (yellow globules and thin faded-green crusts) and many encrusting Sponges with transitional colours. Oscar Andreevich Grimm also described Sponges 'of all sorts of shades from pale yellow to bright red' forming 'crusts up to 1.5 cm thick' or having various shapes, 'sometimes as large as a child’s fist'. Sokolova et al. find no distinctions between sequences of sponges with different appearances and insufficient dissimilarity of skeleton features. This allows them to consider the morphs as a single species, Metschnikowia tuberculata.
At the same time, Sokolova et al. observed some tendency for an increasing number of oxeas’ spines in globular Sponges compared with encrusting ones. Some encrusting Sponges have spicules with minute spines, clearly seen under the scanning electron microscope, but not so obvious under a light microscope. Probably, it was this tendency that led Grimm to misidentify some Sponges with smooth spicules as representatives of marine Haplosclerid genus Reniera (now known under the name Haliclona). Władysław Dybowski claimed that Grimm had missed the spines on the spicule surface. Afterward, Vladimir Koltun figured out that Sponges which had been identified by Grimm as Reniera flava have not only smooth spicules (common to Haplosclerids), but also spiny ones. Moreover, Sponges on Grimm’s drawing of Reniera flava are very similar to those encrusting Sponges collected by Sokolova et al. due to their distinctive, regularly distributed oscula. Thus, Sokolova et al. suggest that Grimm dealt with encrusting Metschnikowia tuberculata having reduced numbers of spines, not with some other Sponge.
The Caspian fauna is considered to be in the process of formation because of significant morphological variety of Fish and benthic Animals, abundance of closely related species with transitional forms, wide ecological niches of species and low specialisation and competitiveness compared with saltwater species (in the Black and Azov Seas). The first two features are also attributable to Sponges of another ancient lake, the Baikal. Being relatively young, its endemic Sponges (family Lubomirskiidae) exhibit a large number of transitional morphological forms between species and possess overlapping morpho-traits. Thus, the morphological variety of studied Metschnikowia tuberculata seems less surprising.
Comparison of Sokolova et al.'s specimens with the museum collection leaves no doubt that they represent Metschnikowia tuberculata. However, spicules of some sponges from the slide collection stand out from others due to their large size and salient spines. Although freshwater Sponges (Spongillida) are known for some spicule variability, at present Sokolova et al. cannot reveal the limits of variability in Metschnikowia. To determine the true diversity of the Caspian Sponges more investigations of specimens from different locations and depths are required.
The current study is based on too few specimens and Sokolova et al. certainly cannot claim all the Caspian Sponges belong to the one species. Nevertheless, our results revealed that sponges of different morphs have identical ITS sequences. Taking into account that ITS sequences have a good resolution at species and generic levels in Spongillida, Sokolova et al. conclude that all the studied samples belong to the one species, Metschnikowia tuberculata. Genetic distances show that Metschnikowia tuberculata clearly belongs to Spongillida.
Sokolova et al.'s data also support the monophyly of Freshwater Sponges previously predicted by morphological data and confirmed by molecular data. The Spongillida are descended from marine Sponges, and have colonised fresh waters, probably through the coastal brackish waters. However, some species remain tolerant of slight salinity. For example, Spongilla alba is apparently associated with brackish waterbodies. Another instance of salinity tolerance occurs in the widespread Sponge Ephydatia fluviatilis, which can survive in mineralised waters like the Baltic Sea and Lake Issyk-Kul.
Thus, the clustering of Metschnikowia with Ephydatia seems unsurprising. This clade also supports the hypothesis of the formation of endemic species from cosmopolitan founders. The high variability of the ITS spacers makes it difficult to align them unambiguously, which leads to low support for deeper nodes. Therefore, more markers are required for improving resolution of the trees.
The CO1 sequences of Freshwater Sponges, conversely, have low variability that resulted in an unresolved phylogeny within Spongillida both in Sokolova et al.'s data and in previous analyses. The CO1 sequence ofMetschnikowia tuberculata is, however, distinct from the Ephydatia fluviatilis CO1 haplotype. These data together with ITS data and morphological differences support the separate taxonomic status of the Sponges.
Spongillidae is shown to be paraphyletic with respect to the Malawispongiid Cortispongilla barroisi. Sokolova et al.'s results support the monophyly of Lubomirskiidae, the sponges of Lake Baikal. They form a strongly supported monophyletic clade with Ephydatia muelleri as a sister species, which is consistent with preceding analyses. However, the genus Radiospongilla turns out to be paraphyletic. Except for this, the obtained results are in accordance with the previous data. They demonstrate the unresolved phylogeny of Spongillida at the family level. Sokolova et al. believe the revision of the taxonomy of Spongillida at the family level requires the addition of other molecular markers and genomic data.
See also...
Follow Sciency Thoughts on Facebook.